|
11/30/2023 0 Comments Iso 13485 quality manual 
In ISO 9001:2015, any clause may be excluded. First, you must define the scope of the quality management system, including details of and justification for any exclusion or non-application. 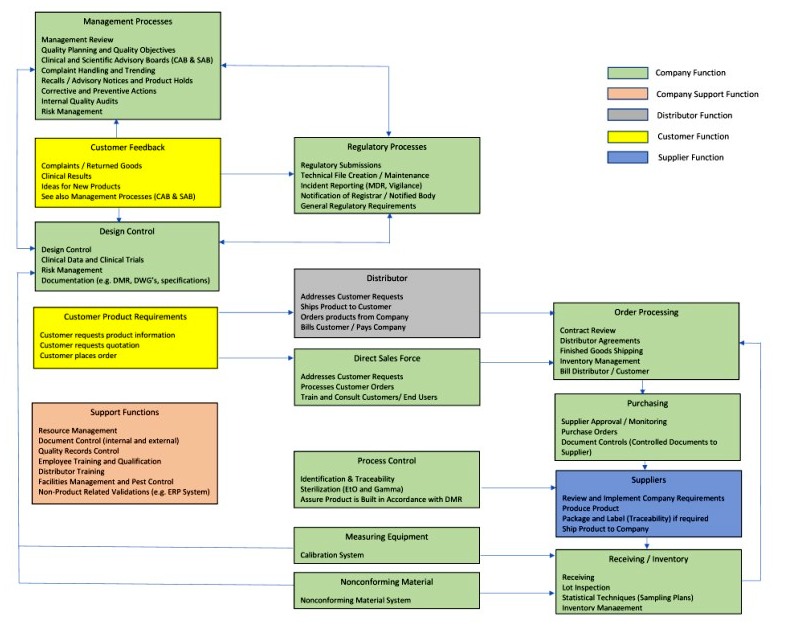
There are four requirements for the content of a quality manual in that sub-clause. It is the primary document meeting the applicable regulatory requirements for a Quality Manual as defined in clause 4.2.2 of ISO 13485:2016. POL-001 is Medical Device Academy’s template for a quality manual. This requirement is found in clause 4.2.1(b). Unlike ISO 9001:2015, the ISO 13485:2016 standard requires that manufacturers establish a quality manual.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
